 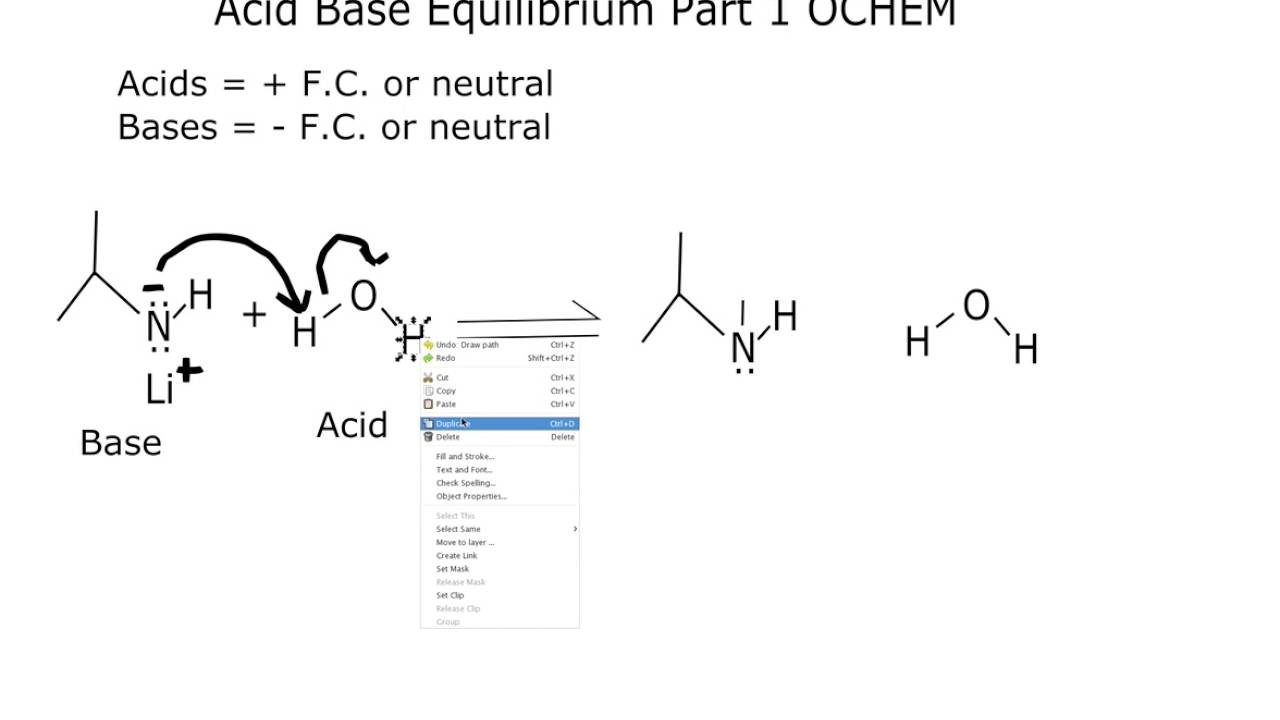
The positive ion from a base can form a salt with the negative ion from an acid. The universal aqueous acid-base definition of the Arrhenius concept is described as the formation of water from hydrogen and hydroxide ions, or hydronium ions and hydroxide ions produced from the dissociation of an acid and base in aqueous solution (2 H 2O → OH - + H 3O + ), which leads to the definition that in Arrhenius acid-base reactions, a salt and water is formed from the reaction between an acid and a base- in more simple scientific definitions, this form of reaction is called a Neutralization reaction. More recent IUPAC recommendations now suggest the newer term "hydronium" be used in favor of the older accepted term "oxonium" to illustrate reaction mechanisms such as those defined in the Brønsted-Lowry and solvent system definitions more clearly, with the Arrhenius definition serving as a simple general outline of acid-base character More succinctly, the Arrhenius definition can be surmised as, "Arrhenius acids form hydrogen ions in aqueous solution with Arrhenius bases forming hydroxide ions." 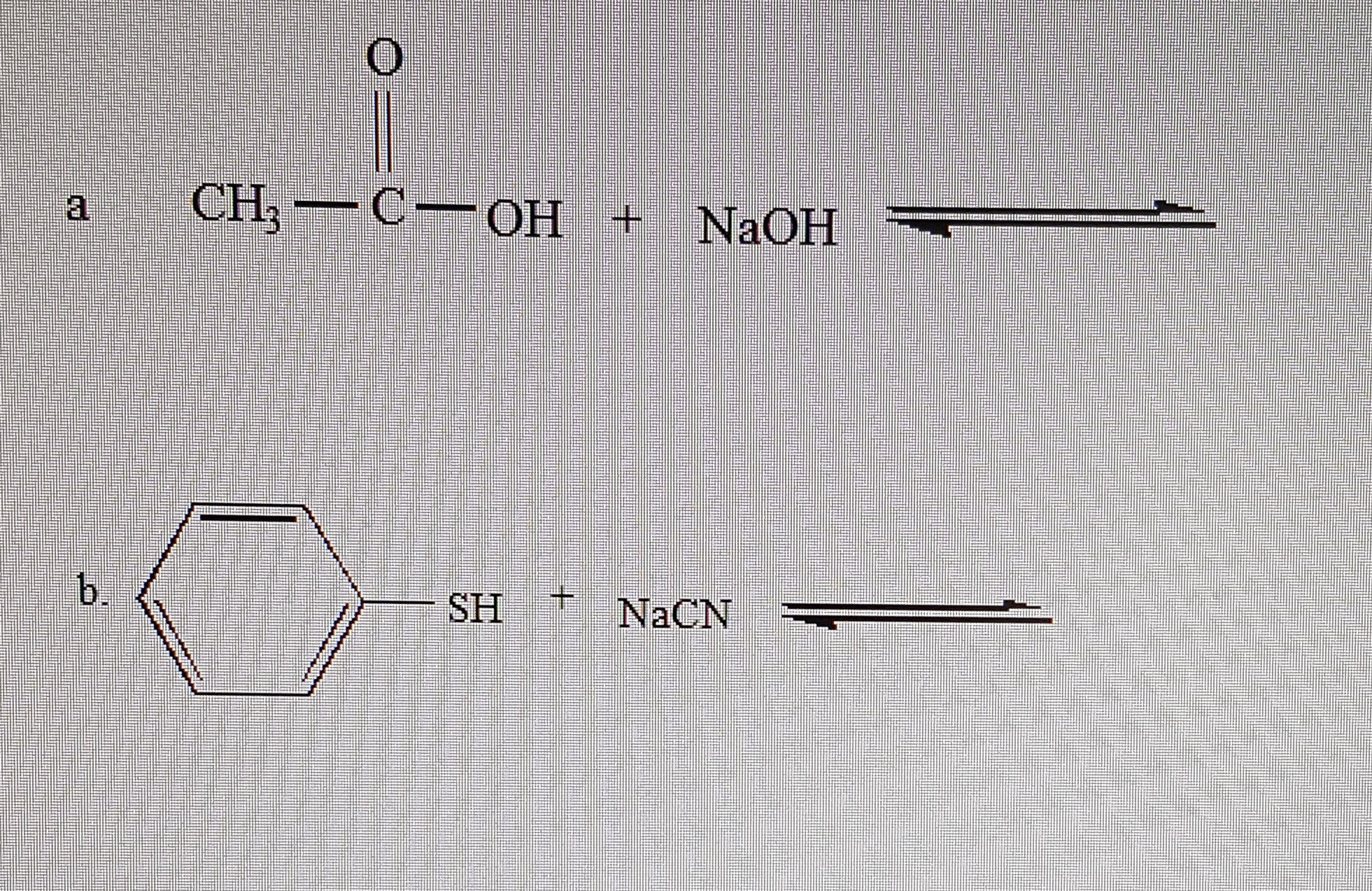 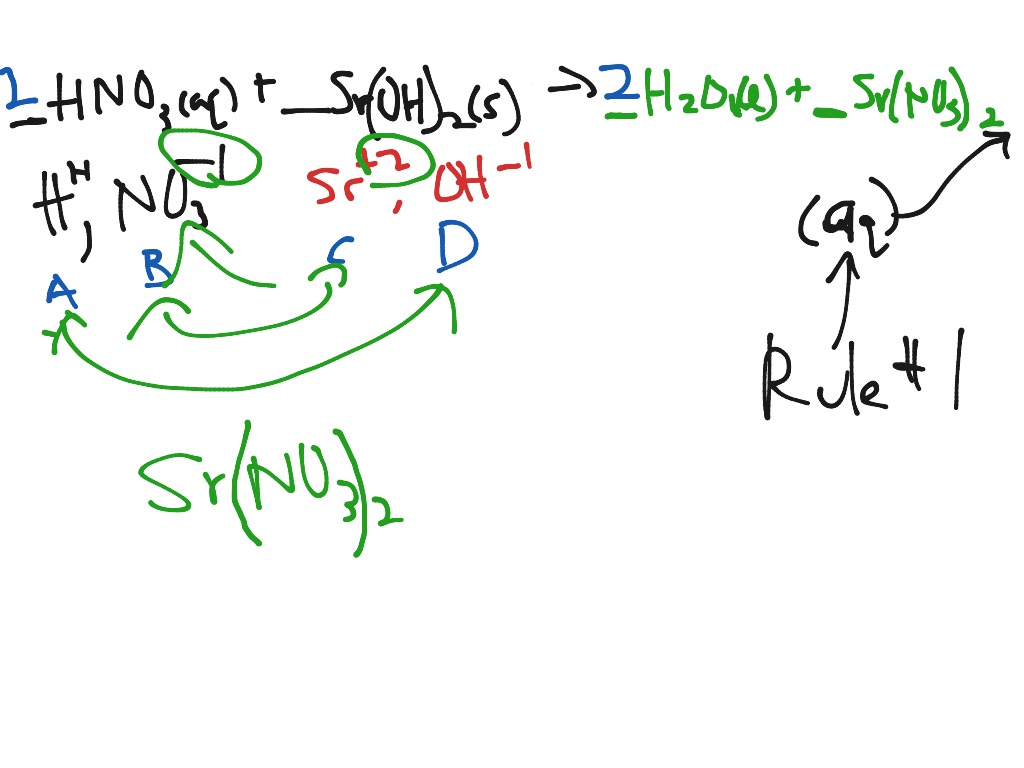
Īs defined at the time of discovery, acid-base reactions are characterized by Arrhenius acids, which dissociate in aqueous solution form hydrogen or the later-termed oxonium (H 3O +) ions, and Arrhenius bases which form hydroxide (OH -) ions. The Arrhenius definition of acid-base reactions is a more simplified acid-base concept devised by Svante Arrhenius, which was used to provide a modern definition of bases that followed from his work with Friedrich Wilhelm Ostwald in establishing the presence of ions in aqueous solution in 1884, and led to Arrhenius receiving the Nobel Prize in Chemistry in 1903 for "recognition of the extraordinary services … rendered to the advancement of chemistry by his electrolytic theory of dissociation". Arrhenius definitionĭevised after the metal-replacement theory of Justus von Liebig and despite some modifications by later theories, the Arrhenius (pictured) concept remains a simple scientific definition of acid-base reaction character. Liebig's definition, while completely empirical, remained in use for almost 50 years until the adoption of the Arrhenius definition. According to Liebig, an acid is a hydrogen-containing substance in which the hydrogen could be replaced by a metal. This finished the doctrinal shift from oxygen-based acids to hydrogen-based acids, started by Davy. The Lavoisier definition was held as absolute truth for over 30 years, until the 1810 article and subsequent lectures by Sir Humphry Davy, in which Davy proved the lack of oxygen in hydrogen sulfide (H 2S), hydrogen telluride (H 2Te), and the hydrohalic acids.Īround 1838, Justus von Liebig proposed a new definition of acids, based on his extensive works on the chemical composition of organic acids. From his limited knowledge, he defined acids in terms of their content of oxygen, and he named oxygen from Greek words meaning "acid-former" (from the Greek οξυς (oxys) meaning "acid" or "sharp" and γεινομαι (geinomai) or "engender"). Lavoisier's knowledge of strong acids was mainly restricted to oxyacids, which tend to contain central atoms in high oxidation states surrounded by oxygen, such as HNO 3 and H 2SO 4, and he was not aware of the true composition of the hydrohalic acids, HCl, HBr, and HI. Historically, the first of the scientific concepts of acids and bases was provided by the French chemist Antoine Lavoisier, circa 1776. Common acid-base theories Lavoisier definition 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
